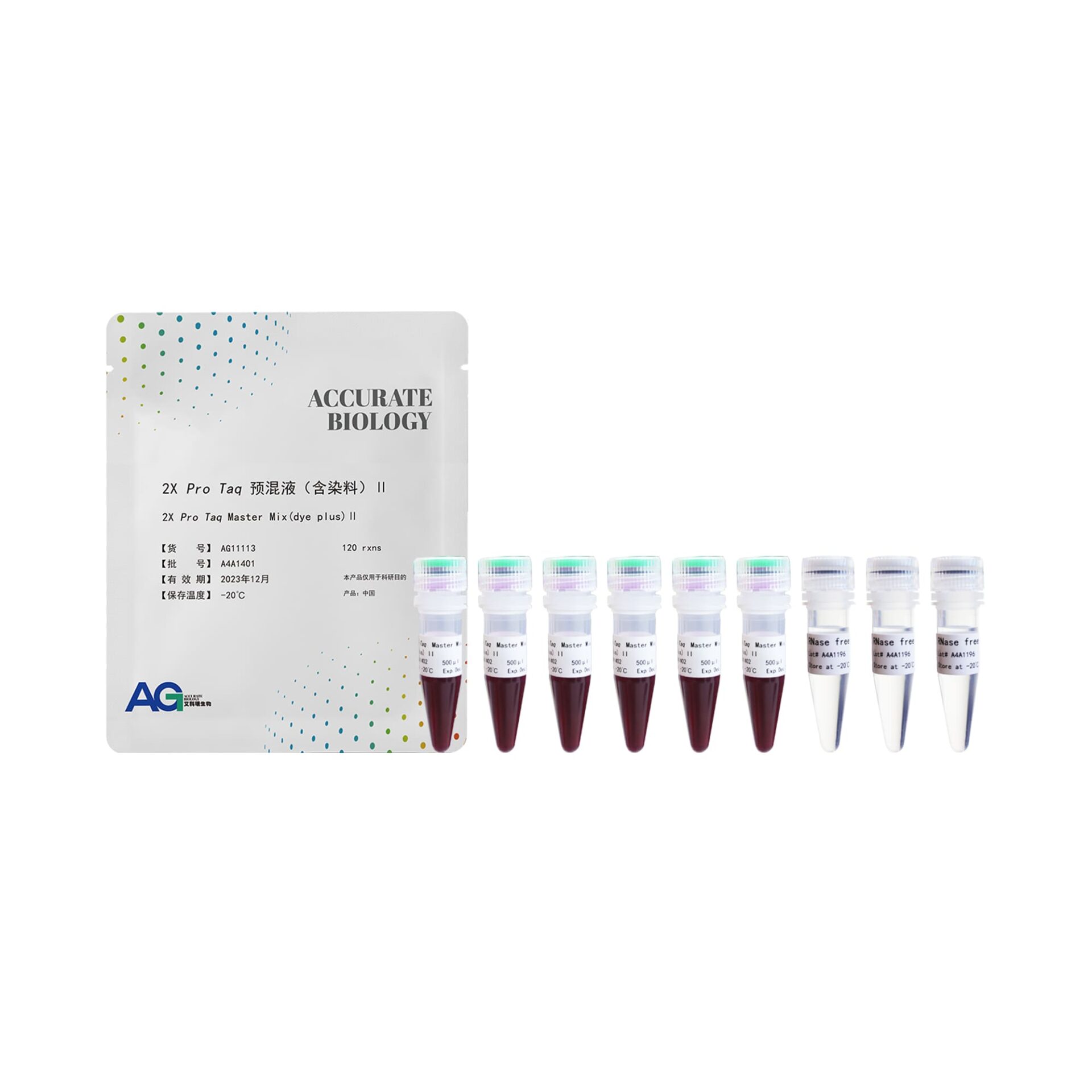
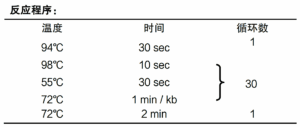
Pro Taq DNA Polymerase is synthesized on our superior performance Accurate Taq High fidelity enzyme has been added to the enzyme to provide 3′→5′ Exonuclease activity (Proof reading activity), which is ideal for amplification of DNA fragments over 10 kb and has good fidelity. This product is a 2-fold concentration PCR reaction premix, containing Pro Taq DNA Polymerase, dNTPs and optimized Buffer system, PCR reaction, just add the template, primers and water to the premixed solution can be amplified. At the same time, the product is also added with the coloring reagent required for electrophoresis testing, the solution of the product is purple-red in color, and it can be used for agarose gel electrophoresis directly after the PCR reaction is completed. The premixed solution is easy to use, minimizes human error, reduces the risk of multi-step contamination, and provides results in a short period of time. The PCR product has an A base at the 3’ end and can be cloned directly into T vectors.
① The product is a 2X premix, only need to add template, primer and water to the premix for amplification, easy to operate, can minimize human error, reduce the possible contamination caused by multi-step operation, and the results can be obtained in a relatively short period of time.
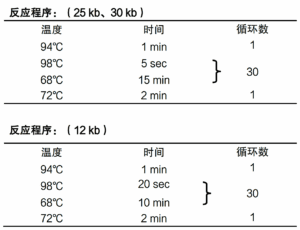
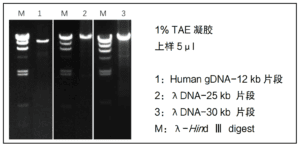
② 2X Pro Taq Master Mix (dye plus) II has good amplification performance, amplifying ~30 kb of DNA fragments using λDNA as a template and ~12 kb of DNA fragments using Human genomic DNA as a template.
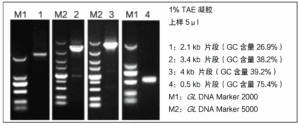
③ The product contains violet dye, after PCR can be directly carried out agarose gel electrophoresis, without adding electrophoresis sample buffer, electrophoresis has a light violet-red indicator band.
| individual parts making up a compound | norm |
| 2X Pro Taq Master Mix (dye plus) II | 500 μl x 6 pc |
| RNase free water | 1 ml x 3 pc |
Transportation temperature: dry ice transportation or -20℃ ice bag transportation